A López-Márquez, C Badosa, L Enjuanes-Ruiz, P Hernández-Carabias, M Sánchez-Martín, B Cadot, Z Guesmia, I Georvasilis, S Balsells, M Blanco-Ramos, E Puighermanal, A Quintana, M Roldán, V Allamand, C Jiménez-Mallebrera. Col6a1 knock-in mice provide a promising pre-clinical model for collagen VI-related dystrophies. Dis Mol Mech, Jan 2026. https://doi.org/10.1242/dmm.052460
Mekzine, L ; Pinzón, N ; Mamchaoui, K ; Kondili, M ; Cadot, B ; Bitoun, M ; Trochet, D. Allele-specific RNAi therapy corrects an extracellular matrix defect in Schuurs-Hoeijmakers syndrome. Am J Hum Genet Oct 2025. DOI10.1016/j.ajhg.2025.07.010.
Chassagne J, Da Silva N, Akrouf I, Cadot B, Julien L, Barthélémy I, Blot S, Le Guiner C, Bui MT, Romero NB, Lainé J, Pietri-Rouxel F, Meunier P, Mamchaoui K, Lorain S, Bitoun M, Benkhelifa-Ziyyat S. Early Endosome Disturbance and Endolysosomal Pathway Dysfunction in Duchenne Muscular Dystrophy. Am J Pathol 2025 https://doi.org/10.1016/j.ajpath.2025.05.007
Traoré M, Noviello C, Vergnol A, Gentil C, Halliez M, Saillard L, Gelin M, Forand A, Lemaitre M, Guesmia Z, Cadot B, Caldas E, Marty B, Mougenot N, Messéant J, Strochlic L, Sadoine J, Slimani L, Jolly A, De la Grange P, Hogrel JY, Pietri-Rouxel F, Falcone S. GDF5 as a rejuvenating treatment for age-related neuromuscular failure. Brain April 2024 https://doi.org/10.1093/brain/awae107
E Lemerle, J Lainé, M Benoist, G Moulay, A Bigot, C Labasse, A Madelaine, A Canette, P Aubin, JM Vallat , N B Romero, M Bitoun, V Mouly, I Marty, B Cadot, L Picas , S Vassilopoulos. Caveolae and Bin1 form ring-shaped platforms for T-tubule initiation. eLife Apr 2023. https://doi.org/10.7554/eLife.84139
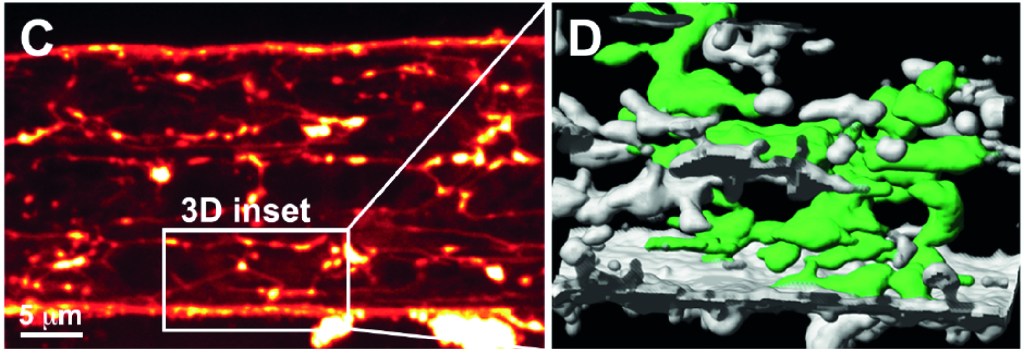
N Rose, B Estrada Chavez, S Sonam, T Nguyen, G Grenci, A Bigot, A Muchir, B Ladoux, B Cadot, F Le Grand, L Trichet. Bioengineering a miniaturized in vitro 3D myotube contraction monitoring chip to model muscular dystrophies. BioMaterials Dec 2022. https://doi.org/10.1016/j.biomaterials.2022.121935

C Le Dour, M Chatzifrangkeskou, C Macquart, M M. Magiera, C Peccate, C Jouve, L Virtanen, T Heliö, K Aalto-Setälä, S Crasto, B Cadot, D Cardoso, N Mougenot, D Adesse, E Di Pasquale, J-S Hulot, P Taimen, C Janke & A Muchir. Actin-microtubule cytoskeletal interplay mediated by MRTF-A/SRF signaling promotes dilated cardiomyopathy caused by LMNA mutations. Nat Comm 2022. https://doi.org/10.1038/s41467-022-35639-x
E Lemerle, J Lainé, G Moulay, A Bigot, C Labasse, A Madelaine, A Canette, P Aubin Tessier, J- Vallat, N Romero, M Bitoun, V Mouly, I Marty, B Cadot, L Picas, S Vassilopoulos. Caveolae and Bin1 form ring-shaped platforms for T-tubule initiation. bioRxiv 2022. https://doi.org/10.1101/2022.11.01.514746
C Meyer, N Beatriz Romero, T Evangelista, B Cadot, J Laporte, A Jeannin-Girardon, P Collet, K Chennen, O Poch. IMPatienT: an Integrated web application to digitize, process and explore Multimodal PATIENt daTa. bioRxiv 2022. https://doi.org/10.1101/2022.04.08.487635
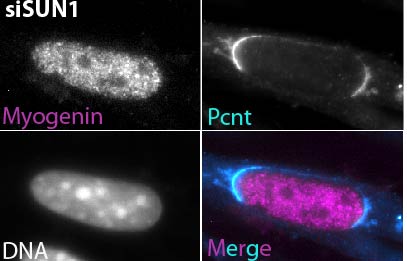
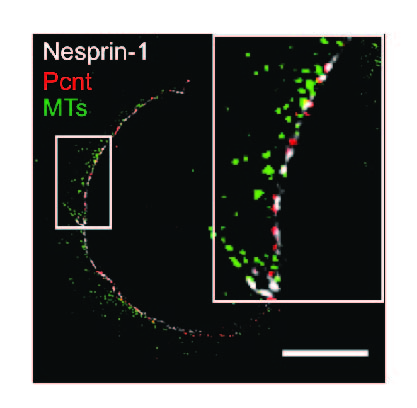
Ei Leen Leong, Nyein Thet Khaing, Bruno Cadot, Wei Liang Hong, Serguei Kozlov, Hendrikje Werner, Esther Sook Min Wong, Colin L Stewart, Brian Burke, Yin Loon Lee. Nesprin-1 LINC complexes recruit microtubule cytoskeleton proteins and drive pathology in Lmna mutant striated muscle. Human Molecular Genetics (2022). https://doi.org/10.1093/hmg/ddac179
Dudhal S, Mekzine L, Prudhon B, Soocheta K, Cadot B, Mamchaoui K, Trochet D, Bitoun M. Development of versatile allele-specific siRNAs able to silence all the dominant Dynamin 2 mutations. Mol Ther Nucleic Acids 2022. Development of versatile allele-specific siRNAs able to silence all the dominant Dynamin 2 mutations. Mol Ther Nucleic Acids 2022.
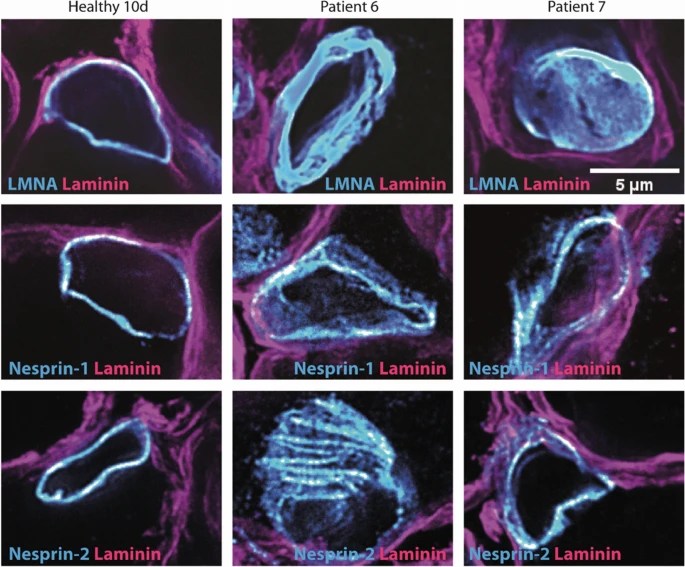
Labasse, C., Brochier, G., Taratuto, AL., Cadot B., Rendu J., Monges S., Biancalana V., Quijano‑Roy S., Thao Bui M., Chanut A., Madelaine A, Lacène E., Beuvin M, Amthor H., Servais L., de Feraudy Y, Erro M., Saccoliti M., Abath Neto O., Fauré J., Lannes B., Lauge V., Coppens S, Lubieniecki F, Buj Bello A, Laing N., Evangelista T, Laporte J, Böhm J. and Romero NB. Severe ACTA1-related nemaline myopathy: intranuclear rods, cytoplasmic bodies, and enlarged perinuclear space as characteristic pathological features on muscle biopsies. acta neuropathol commun 10, 101 (2022). https://rdcu.be/cRm36
Gökçe Agsu, Jérémie Gaillard, Bruno Cadot, Laurent Blanchoin, Emmanuelle Fabre, Manuel Théry. Reconstituting the Interaction Between Purified Nuclei and Microtubule Network. In: Inaba, H. (eds) Microtubules. Methods in Molecular Biology, vol 2430. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1983-4_25
Romero N & Cadot B. The Muscle Atlas: a databank of muscle biopsies’ images. Cah. Myol. 2021 ; 23 : 20-23. https://doi.org/10.1051/myolog/202123005
Francisco J. Calero-Cuenca, Daniel S. Osorio, Sofia Carvalho-Marques, Sreerama Chaitanya Sridhara, Luís M. Oliveira, Yue Jiao, Jheimmy Diaz, Cátia S. Janota, B Cadot & E R. Gomes. Ctdnep1 and Eps8L2 regulate dorsal actin cables for nuclear positioning during cell migration.Curr Biol (2021). https://www.sciencedirect.com/science/article/pii/S0960982221000403

Davidson PM & Cadot B. Actin on and around the nucleus. Trends in Cell Biology (2021). https://www.cell.com/trends/cell-biology/fulltext/S0962-8924(20)30244-0
, , , , , , , ,
Déjardin T, Carollo PS, Sipieter F, Davidson PM, Seiler C, Cuvelier D, Cadot B, Sykes C, Gomes ER, Borghi N. Nesprins are mechanotransducers that discriminate Epithelial-Mesenchymal Transition programs. JCB (2020). https://rupress.org/jcb/article/219/10/e201908036/152020

Girardi F, Taleb A, Ebrahimi M, Datye A, Gamage DG, Peccate C, Giordani L, Millay DG, Gilbert PM, Cadot B and Le Grand F. TGFβ signaling curbs cell fusion and muscle regeneration. Nat Comm (2021). https://www.nature.com/articles/s41467-020-20289-8
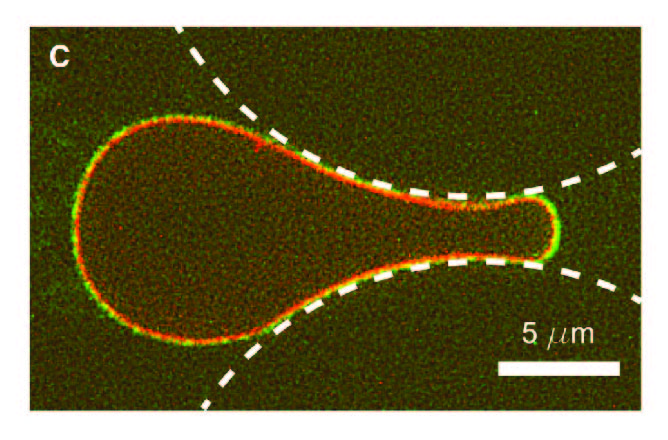
, , , , , , ,
Darrigrand JF, Valente M, Comai G, Martinez P, Petit M, Nishinakamura R, Osorio DS, Renault G, Marchiol C, Ribes V, Cadot B. Dullard-mediated Smad1/5/8 inhibition controls mouse cardiac neural crest cells condensation and outflow tract septation. eLife. Feb 2020. https://doi.org/10.7554/eLife.50325


M Traoré, C Gentil, C Benedetto, JY Hogrel, P De la Grange, B Cadot, S Benkhelifa-Ziyyat, L Julien, M Lemaitre, A Ferry, F Piétri-Rouxel, and S Falcone. An embryonic CaVβ1 isoform promotes muscle mass maintenance via GDF5 signaling in adult mouse. Science Translational Medecine. Nov 2019. https://stm.sciencemag.org/content/11/517/eaaw1131
Cadot B. La différenciation des cellules musculaires striées squelettiques. Planet-Vie, Jeudi 28 juin 2018 Link
Cadot B. Editorial: Nuclear positioning: a matter of life. Semin Cell Dev Biol (2017), https://doi.org/10.1016/j.semcdb.2017.11.034. ⇒
Gimpel P, Lee YL, Sobota RM, Calvi A, Koullourou V, Patel R, Mamchaoui K, Nédélec F, Shackleton S, Schmoranzer J, Burke B, Gomes ER, Cadot B. Nesprin-1α-Dependent Microtubule Nucleation from the Nuclear Envelope via Akap450 Is Necessary for Nuclear Positioning in Muscle Cells. Current Biology. 2017 Sep 27 pii: S0960-9822(17)31069-2. doi: 10.1016/j.cub.2017.08.031. Link

Roman W, Martins JP, Carvalho FA, Voituriez R, Abella JVG, Santos NC, Cadot B, Way M, Gomes ER. Myofibril contraction and crosslinking drive nuclear movement to the periphery of skeletal muscle. Nature Cell Biology. 2017 Sep 11. doi: 10.1038/ncb3605. Link
Gomes ER & Cadot B. Molecular Motors and Nuclear Movements in Muscle. Article Addendum. Communicative & Integrative Biology. 2017. Link
Gache V, Gomes ER, Cadot B. Molecular motors involved in nuclear movement during skeletal muscle differentiation. Molecular Biology of the Cell. April 1, 2017 vol. 28 no. 7 865-874. Link

Pimentel M., Falcone S., Cadot B., Gomes ER. Isolation of Mouse Myoblasts for Differentiation and Imaging of Mature Myofibers. J. Vis. Exp. 2016 e55141, doi:10.3791/55141. Link .
Vilmont V., Cadot B., Ouanounou G, Schmitt A, Gomes ER. A system for studying mechanisms of neuromuscular junction development and maintenance. Development. 2016 Jul 1;143(13):2464-77. Link
Vilmont V., Cadot B., Vezin E, Le Grand F, Gomes ER. Dynein disruption perturbs post-synaptic components and contributes to impaired MuSK clustering at the NMJ: implication in ALS. Scientific Reports. 2016 Jun 10;6:27804. Link
Cadot B, Gache V, Gomes ER. Moving and positioning the nucleus in skeletal muscle – one step at a time. Nucleus. 2015 Sept link. (Corresponding author) Link
Zalc A., Rattenbach R., Auradé F., Cadot B., Relaix F. Pax3 and Pax7 play essential safeguard functions against environmental stress-induced birth defects. Developmental Cell. 2015 Apr 6;33(1):56-66. Link
Cadot B, Gomes ER. Skeletal muscle. Encyclopedia of Cell Biology. 2016 Elsevier, Pages 677–682 Link
Cadot B, Gache V, Gomes ER. Fast, multi-dimensional and Simultaneous Kymograph-like Particle dynamics (SkyPad) analysis. PlosOne. 2014 Feb 19;9(2):e89073. (Corresponding author) Link
Cadot B, Gache V, Gomes ER. Spatial distribution of nuclei in muscle fibers: a novel actor of muscular function. Med Sci (Paris). 2012 Jun-Jul;28(6-7):577-9. Link
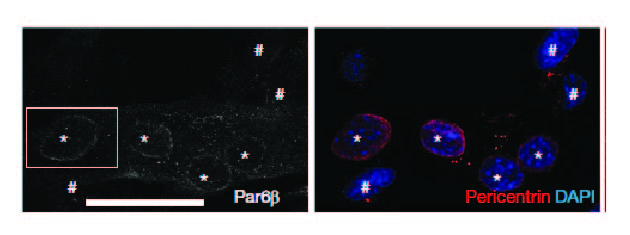
Cadot B, Gache V, Vasyutina E, Falcone S, Birchmeier C, Gomes ER. Nuclear movement during myotube formation is microtubule and dynein dependent and is regulated by Cdc42, Par6 and Par3. EMBO Rep. 2012 Aug 1;13(8):741-9. Link
Metzger T, Gache V, Xu M, Cadot B, Folker ES, Richardson BE, Gomes ER, Baylies MK. MAP and kinesin-dependent nuclear positioning is required for skeletal muscle function. Nature. 2012 Mar 18;484(7392):120-4. Link
Rufini S, Lena AM, Cadot B, Mele S, Amelio I, Terrinoni A, Desideri A, Melino G, Candi E. The sterile alpha-motif (SAM) domain of p63 binds in vitro monoasialoganglioside (GM1) micelles. Biochem Pharmacol. 2011 Nov 15;82(10):1262-8. doi: 10.1016/j.bcp.2011.07.087.
Cadot B., Brunetti B., Coppari S., Fedeli S., de Rinaldis E, Claudio dello Russo, Gallinari P., De Francesco R., Steinkühler C. and Filocamo G. Loss of HDAC4 causes segregation defects during mitosis of p53-deficient human tumor cells. Cancer Research. 2009;69(15):6074-82.
Cicero, DO., Falconi, M., Candi, E., Mele, S., Cadot, B., Di Venere, A., Rufini, S., Melino, G., Desideri (2006). NMR structure of the p63 SAM domain and dynamic properties of G534V and T537P pathological mutants. Cell Biochemistry and Biophysics. 2006;44(3):475-89.
Cadot, B., Rufini, A., Pietroni, V., Ramadan, S., Guerrieri, P., Melino, G., Candi, E. (2004) Overexpressed transglutaminase 5 triggers cell death. Amino Acids. Jul;26(4):405-8. Cited 3 times.
Terrinoni, A., Ranalli, M., Cadot, B., Leta, A., Bagetta, G., Vousden, K.H., Melino, G. (2004) p73-alpha is capable of inducing scotin and ER stress. Oncogene. Apr 29;23(20):3721-5.
Candi, E., Paradisi, A., Terrinoni, A., Pietroni, V., Oddi, S., Cadot, B., Jogini, V., Meiyappan, M., Clardy, J., Finazzi-Agro, A., Melino, G. (2004) Transglutaminase 5 is regulated by guanine-adenine nucleotides. Biochem J. Jul 1;381(Pt 1):313-9. Cited 62 times.
Candi E., Paradisi A., Terrinoni A., Cadot B., Rufini A., Puddu P., Melino G. (2002) Role of Transglutaminase 5 in epidermis. Minerva Biotec. 14: 155-158.
Glukhova, L., Angevin, E., Lavialle, C., Cadot, B., Terrier-Lacombe, M.J., Perbal, B., Bernheim, A., Goguel, A.F. (2001) Patterns of specific genomic alterations associated with poor prognosis in high-grade renal cell carcinomas. Cancer Genet Cytogenet. Oct 15;130(2):105-10.
Cadot, B., Maillard, M., Ball, R.Y., Sethia, K., Edwards, D.R., Perbal, B., Tatoud, R. (2001) Differential expression of the ccn3 (nov) proto-oncogene in human prostate cell lines and tissues. Mol Pathol. Aug;54(4):275-80.
